|
Design Parameters for a Natural Uranium Fueled Nuclear Reactor, C. Uranium is a naturally occurring radioactive element, which has the atomic number of 92 and corresponds to the chemical symbol U in the periodic table.Similarly, enriched uranium was referred to as Oralloy ( Oak Ridge alloy), and depleted uranium was referred to as Depletalloy (depleted alloy). Uranium was also codenamed "X-Metal" during World War II. Therefore, a billion years ago, there was more than double the uranium-235 compared to now.ĭuring the Manhattan Project, the name Tuballoy was used to refer to natural uranium in the refined condition this term is still in occasional use. Uranium-235 decays at a faster rate ( half-life of 700 million years) compared to uranium-238, which decays extremely slowly (half-life of 4.5 billion years). On rare occasions, earlier in geologic history when uranium-235 was more abundant, uranium ore was found to have naturally engaged in fission, forming natural nuclear fission reactors. Unenriched natural uranium is appropriate fuel for a heavy-water reactor, like a CANDU reactor. In its refined state, it is a heavy, silvery-white metal that is malleable, ductile, slightly paramagnetic, and. Uranium is an actinide element, and has the highest atomic mass of any naturally occurring element. How many neutrons are in the nuclei of each of these. Advanced Note: This atomic mass is slightly higher than the value given in the periodic table for the element carbon. For the final answer, add these together: 11.76 + 0.26 12.02 g/mol. It is more plentiful than silver and about as abundant as molybdenum or arsenic. Three isotopes of uranium have mass numbers of 234, 235, and 238. Next, multiply the atomic mass of each isotope by the proportion of the element in the sample: 0.98 x 12 11.76. Nuclear weapons take a concentration of 90% uranium-235, and light water reactors require a concentration of roughly 3% uranium-235. Uranium is a naturally occurring element that makes up approximately 24 ppm of the earths crust. 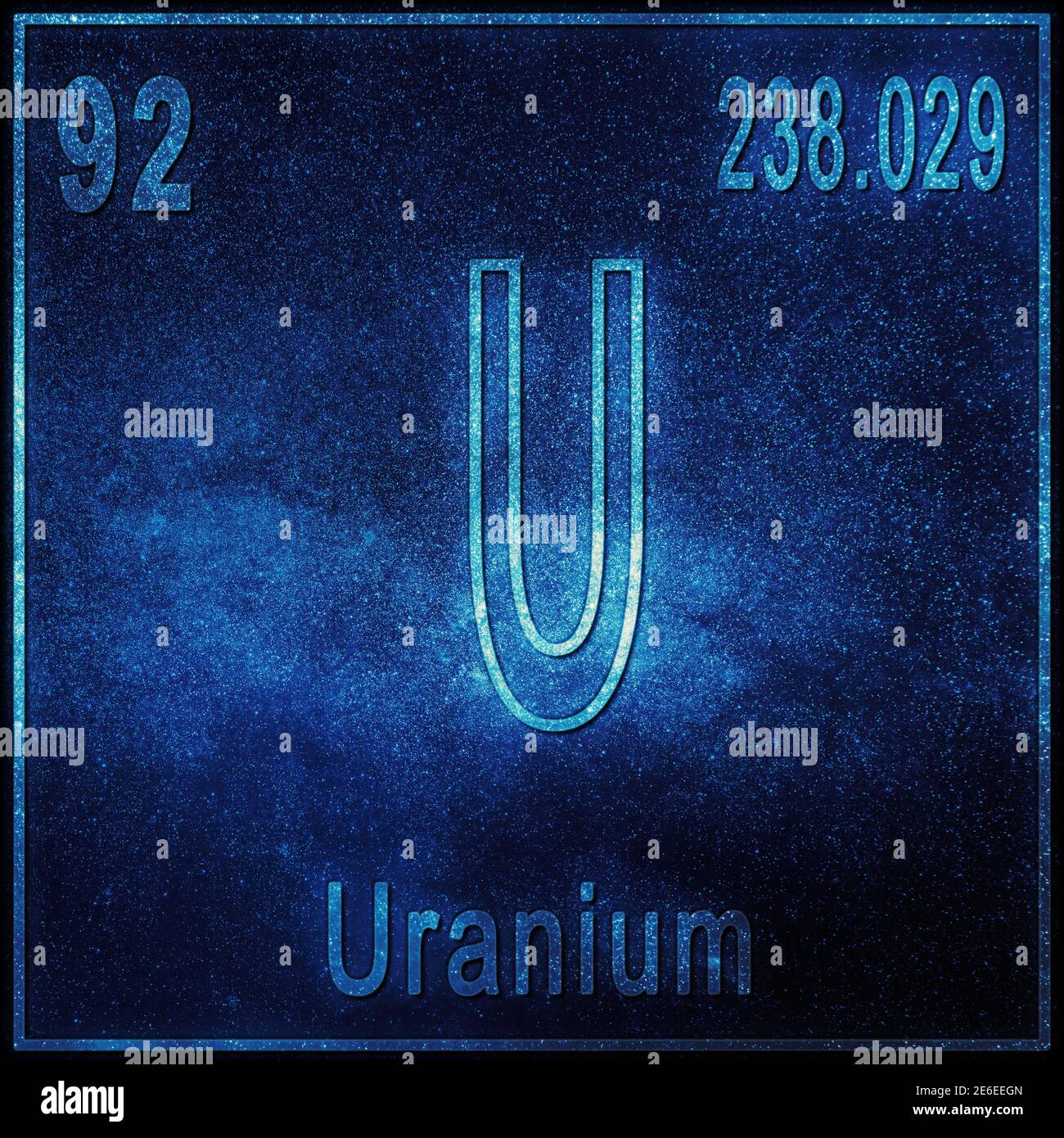
The 0.72% uranium-235 is not sufficient to produce a self-sustaining critical chain reaction in light water reactors or nuclear weapons these applications must use enriched uranium. However, experimental fuelings with uranium trioxide (UO 3) and triuranium octaoxide (U 3O 8) have shown promise. Historically, graphite-moderated reactors and heavy water-moderated reactors have been fueled with natural uranium in the pure metal (U) or uranium dioxide (UO 2) ceramic forms. Natural uranium can be used to fuel both low- and high-power nuclear reactors. Approximately 2.2% of its radioactivity comes from uranium-235, 48.6% from uranium-238, and 49.2% from uranium-234. Natural uranium ( NU or U nat ) is uranium with the same isotopic ratio as found in nature. JSTOR ( March 2013) ( Learn how and when to remove this template message).Unsourced material may be challenged and removed. 
Please help improve this article by adding citations to reliable sources. This is fortunate for atomic power generation, for without this delay 'going critical' would be an immediately catastrophic event. These stars, which often form as pairs in the aftermath of a supernova – can eventually collide with one another.This article needs additional citations for verification. The critical mass for lower-grade uranium depends strongly on the grade: with 20 235 U it is over 400 kg with 15 235 U. Uranium (atomic number 92 molar mass 238.03 g/mol) has the largest molar mass of any element naturally found on. 
This process unfolds during neutron star events. Elements with masses greater than 260, are forged via rapid neutron capture, or ‘r-process’, which produces most of the elements that are heavier than iron (atomic mass 55.8), including uranium, platinum, gold and silver. Most heavier elements with masses ranging from 237 (Neptunium) up to 266 (Lawrencium) are the result of human processes or experimentation.īut neutron stars have long been explored as veritable heavy element factories. With an atomic mass of 238, uranium is the heaviest naturally occurring element known on Earth, though others like plutonium have been found in trace amounts due to reactions in uranium deposits. 
1 Da is defined as 112 of the mass of a free carbon-12 atom at rest in its ground state. Although the SI unit of mass is the kilogram (symbol: kg), atomic mass is often expressed in the non-SI unit dalton (symbol: Da) equivalently, unified atomic mass unit (u). Move over uranium, the Milky Way’s oldest stars have bigger and better elements to make.Ī group of researchers from across the United States, Canada and Sweden have discovered ancient neutron stars might have created elements with atomic mass greater than 260. The atomic mass ( ma or m) is the mass of an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
